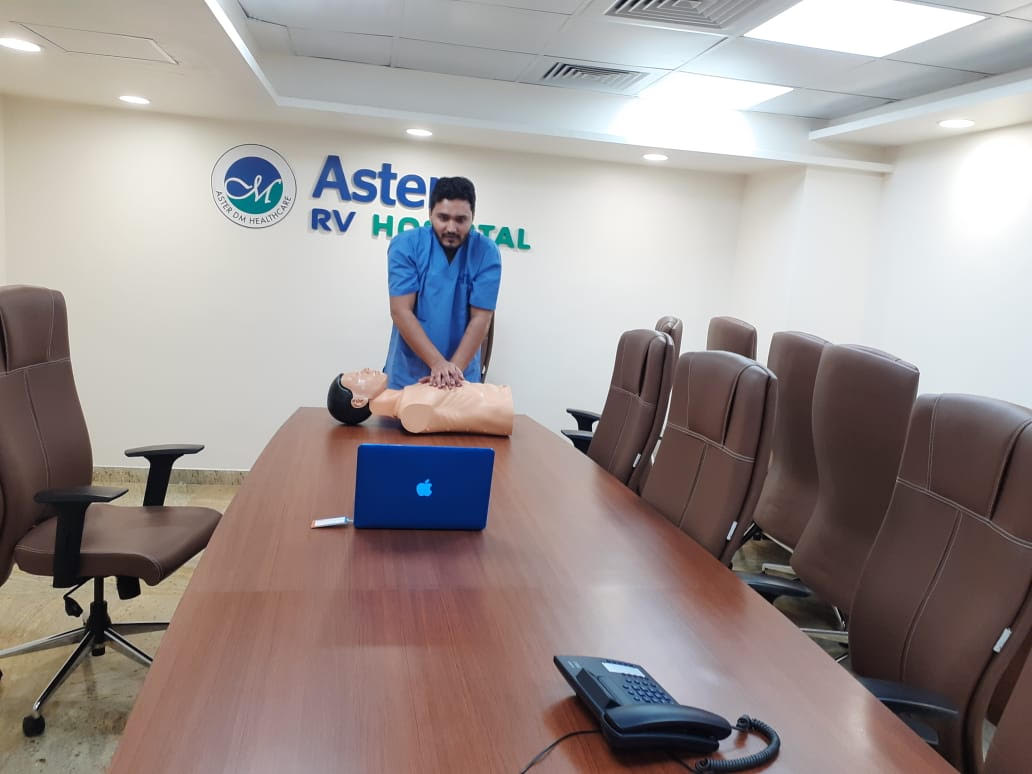
The Central Government has taken due cognisance of the contributions made by the Medical Devices segment to the overall healthcare industry in general and the Covid period in particular. The Government has made specific declarations pertaining to the industry, and many of these are in line with recommendations that we have been making to the Government. All these developments will promote the medical devices industry and create a favourable business environment
ICMR Labs across the country will be made available to the corporate and manufacturing sector for the development of Medtech products. This will specially be beneficial for the Medtech start-up ecosystem which is cash-strapped and may not have adequate resources to set up their own dedicated laboratory infrastructure
Multi-dimensional Medical Devices segment courses will be introduced for the holistic development of all facets of the Medtech segment. This will create a pool of qualified talent resource for the MedTech segment which has been facing a dearth of qualified manpower.
The Government has also announced the setting up of 100 labs for the development of 5G applications. The digital ecosystem is key for the delivery of the MedTech solutions, and MedTech – Telemedicine – Digital Health will majorly benefit from this move.
The Government has announced the removal of 39,000 legal provisions and compliances, and this will certainly contribute to the Ease of Doing Business aspect. We are hopeful that many of these will be pertaining to the healthcare – medical devices segment.
The Government has also announced the reduction of Indirect Taxes / Custom duties from 21 % to 13 %. We are yet to analyse the fine print and actual affected components, but are hopeful that it will address some (if not all) that affect the medical devices segment, especially raw material.
The Government’s announcement promoting startups and increasing the period of carry forward of losses from 7 years to 10 years will positively impact the start-up ecosystem, which includes the healthcare – medical devices segment.
To unleash Innovation and research by Startups and Academia, a National Data Governance Policy will be brought out. This will enable access to anonymized data and will further promote innovation and R&D within industries across board.
The FM has announced the setting up of 3 Centres of Excellence for Artificial Intelligence in top educational institutions. This will drive the make in India initiative, and since the MedTech segment is has AI as a potent growth component, we are hopeful of this driving a positive growth for the medical devices segment as well.
New Schemes are also being announced for the promotion of R&D in the pharmaceutical segment through setting up of Centres of Excellence
We are going through the various aspects of the budget, and will be able to provide a better reaction after analysing all the aspects and the fine-print
Jatin Mahajan
Secretary – Association of Diagnostic Manufacturers of India (ADMI)
Managing Director – J Mitra & Company