EcoStar™ Auto device is a Positive Pressure device indicated for the treatment of Obstructive Sleep Apnea and Hypopnea Syndrome (OSAHS) in patients weighing more than 30 kg (66 lbs). They can be used at home or in a sleep center.
EcoStar™ Auto is a Positive Pressure device with a large range of features to improve the comfort and treatment compliance of patients suffering from Obstructive Sleep Apnea Syndrome (OSAS). This device adapts easily to the user’s daily routine, and guarantees high-performance, comfort and ease of use. With its exceptionally light weight (750 g), compactness and universal power adapter, EcoStar™ Auto optimizes user comfort, without requiring special adjustments, at home or on the move.
The EcoStar™ Auto is designed to be compatible with the humidifier GoodKnight® H2O when the patient’s treatment includes heated humidification.
The air inlet filter is highly accessible, making it easy for the user to independently perform routine maintenance on the device.
EcoStar Auto is equipped with state-of-the-art technologies and algorithms to optimize user comfort and compliance: comfort mode, resistance compensation for any type of mask and tube, intelligent ramp, hands-free start (Auto-on) and Auto-off are standard.
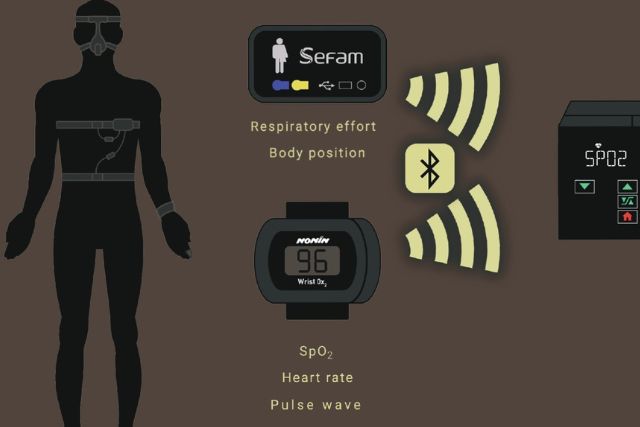
EcoStar™ Auto detects inspiratory flow limitation, snoring and residual obstructive / central apneas and hypopneas. It differentiates between obstructive and central events and allows the device to deliver the optimum pressure at any time.
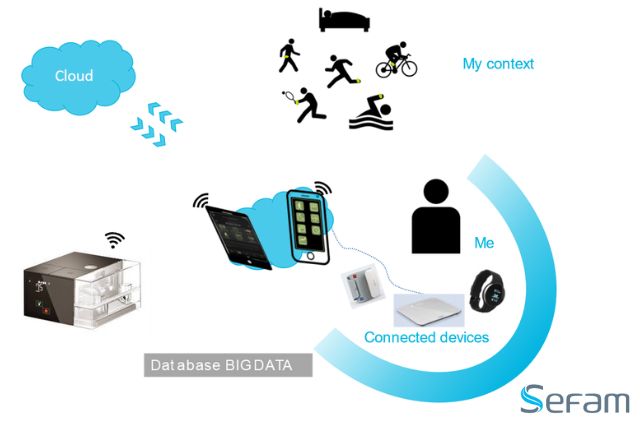
EcoStar Auto saves the efficacy data on leaks and usage for one year in the device’s memory, along with the real patient flow over the last 16 hours of use.
These data are transferred to the DreamStar Analyze software and presented in the form of summary reports and graphs for simple and complete assessment of the treatment efficacy.
EcoStar Auto works with our Oxylink oximetry too.
EcoStar™ Auto features:
Disconnected mask function
If the mask is removed by the patient, the pressure is reduced to the minimum level in a few seconds. The device stops after 30 minutes, without resuming treatment.
Auto ON function
The patient starts his/her treatment either by breathing in the mask or by pressing the on/off button.
Calibration Comfort (CC+) function
This function improves the respiratory comfort of the patient during treatment in PPC and Auto-PPC mode. It adapts the supplied pressure to the patient’s respiratory cycle without compromising the effectiveness of the treatment.
This technology allows the pressure to be increased on inhalation and decreased on exhalation; these variations adapt to the patient’s natural respiratory cycles.
Specific auto-calibration function
This function delivers the prescribed pressure to the patient interface, whatever the configuration of the air pathway to the interface (type of mask, diameter and length of tube, humidifier or not).
Ramp function
This helps the patient to fall asleep with a reduced pressure, called comfort pressure. After a pre-set ramp time, the device delivers the prescribed pressure in PPC mode or activates the pressure increase and decrease commands in the Auto-CPAP mode.
For more information:
Customer Service | India
T: +91 777 8910 601
Customer Service | France
T: +33 (0)3 83 44 85 50
About Sefam:
Since it was founded in 1982, the Nancy-based French company Sefam has been designing, manufacturing, and commercializing diagnostic and therapy devices for Sleep disordered Expertise: Proud of our history and our 30 years of experience, we put all our expertise at the service of our patients. We are constantly looking to improve our solutions, to create even more accurate and personalized cutting-edge technology. Sefam places a great deal of importance on feedback from customers, doctors, distributors and home healthcare providers, because only by working together the perfect solutions can be derived to achieve a common goal: to improve the health of patients. The Medical Advisory Board, guides Sefam. Thus, the products and services are inspired by doctors and researchers. Sefam has all the necessary quality certifications (ISO 9001, EN 13485, FDA, etc.) demonstrating quality and safety of our devices.