trategies to improve lung recruitment in patients with acute respiratory distress syndrome (ARDS) and respiratory failure can include the use of B-agonists. These drugs are used extensively to treat hypoxemic ventilated patients even without a confirmed clinical benefit. A recent study has investigated whether providing inhaled Salbutamol delivered by Aerogen technology can improve the lung function of paediatric patients14. Compared to baseline, aerosolised Salbutamol improved the functional residual capacity of critically ill children with respiratory failure. This study provides new evidence for the use of aerosolised B-agonists as another strategy to improve lung recruitment with ARDS14.
Comparison of Aerogen with MDIs
Although drug delivery efficiency has been shown to be similar between a pressurised metered dose inhaler (pMDI) and Aerogen22, the actual dose emitted from the pMDIs (e.g, 100μg per actuation with Salbutamol) is much lower than the typical 2.5mg dose used with Aerogen. In addition, pMDIs aren’t without difficulties as failure to synchronise actuations with inspiration has been shown to reduce the aerosol drug delivery25. It is also important to ensure canisters are shaken before use as the dose may vary due to separation from the propellant26.
There are several studies, which provide evidence that the cost savings of switching from combivent MDI to the Aerogen Solo is significant15-18, 27.
Blake et al. discussed substantial cost savings in conjunction with staff satisfaction after switching and a potential system wide annual saving of up to $1.74 million across 105 hospitals15.
Loborec et al. investigated the financial impact of replacing ipratropium-albuterol MDIs for Aerogen and calculated a three-month cost saving of $99,359 and projected yearly saving of $397,436.18
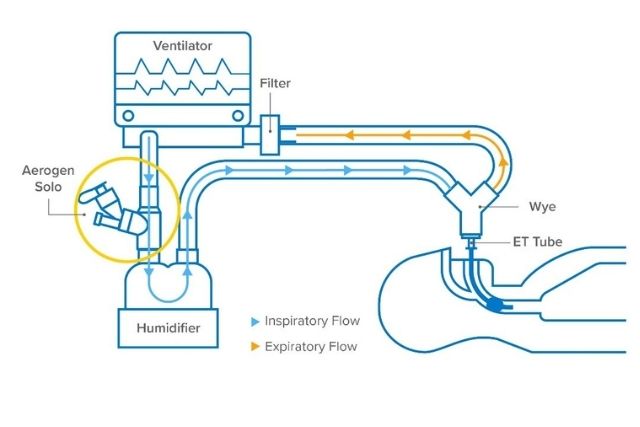
Closed-circuit-with-Aerogen
Mitigating the transmission of patient generated infectious aerosols
The latest GOLD Report1 states that for COVID-19 patients receiving ventilatory support
It is vital to keep the circuit intact and prevent the transmission of the virus. Using a mesh nebulizer in ventilated patients allows adding medication without requiring the circuit to be broken for aerosol drug delivery.
Aerogen is the only closed circuit aerosol drug delivery system that mitigates the transmission of patient generated infectious aerosol2-7 and delivers effective aerosol treatment.2,8,9
Discover how Aerogen’s vibrating mesh technology, can efficiently deliver inhalation treatments for COVID-19 patients and is designed for the safety of both the patient and the caregiver.
Multiple international guidance has been published outlining the use of this mesh technology in the treatment of COVID-19 patients. 1,4,6, 10, 11
Why is a Closed Circuit important?
Prevention of VAP (Ventilator Acquired Pneumonia)/VAE
1.Ventilator-associated pneumonia (VAP) is a nasocomial (hospital acquired) pneumonia that develops more than 48 hours after endotracheal intubation.
2.It is one of the most common infections acquired by adults and children in intensive care units (ICUs) and is a cause of significant patient morbidity and mortality, increased utilisation of healthcare resources, and excess cost.
For further information, please contact:
Kamal Kishore
Business Manager -India
Tel: +91 9971700662
[email protected]
Rajiv Julka
Country Manager -India & Subcontinent
Tel: +919810093138
[email protected]
References:
1.Ari A, Atalay OT, Harwood R, Sheard MM, Aljamhan EA and Fink JB. Influence of nebulizer type, position, and bias flow on aerosol drug
2.delivery in simulated pediatric and adult lung models during mechanical ventilation. Respiratory care. 2010;55:845-51.
3.Berlinski A and Willis JR. Albuterol delivery by 4 different nebulizers placed in 4 different positions in a pediatric ventilator in vitro model. Respiratory care. 2013;58:1124-33.
4.Fang TP, Lin HL, Chiu SH, Wang SH, DiBlasi RM, Tsai YH and Fink JB. Aerosol Delivery Using Jet Nebulizer and Vibrating Mesh Nebulizer During High Frequency Oscillatory Ventilation: An In Vitro Comparison. Journal of aerosol medicine and pulmonary drug delivery. 2016.
5.Abdelrahim ME, Plant P and Chrystyn H. In-vitro characterisation of the nebulised dose during non-invasive ventilation. The Journal of pharmacy and pharmacology. 2010;62:966-72.
6.Galindo-Filho VC, Ramos ME, Rattes CS, Barbosa AK, Brandao DC, Brandao SC, Fink JB and de Andrade AD. Radioaerosol Pulmonary Deposition Using Mesh and Jet Nebulizers During Noninvasive Ventilation in Healthy Subjects. Respiratory care. 2015;60:1238-46.
7.McPeck M. Improved Aerosol Drug Delivery with an Electronic Mesh Nebulizer during Non-invasive Ventilation AARC poster. 2012
8.Ari A, Harwood R, Sheard M, Dailey P and Fink JB. In vitro comparison of heliox and oxygen in aerosol delivery using pediatric high flow nasal cannula. Pediatric pulmonology. 2011;46:795-801.
9.Reminiac F, Vecellio L, Heuze-Vourc’h N, Petitcollin A, Respaud R, Cabrera M, Le Pennec D, Diot P and Ehrmann S. Aerosol Therapy in Adults Receiving High Flow Nasal Cannula Oxygen Therapy. Journal of aerosol medicine and pulmonary drug delivery. 2015.
10.Alcoforado L, de Melo Barcelar J, Castor Galindo V, Cristina S. Brandão S, Fink J, B. and Dornelas de Andrade A. Analysis of Deposition Radioaerosol Nebulizers Membrane in Healthy Subjects. ISAM poster presentation 2015.
11.Ari A, de Andrade A, Sheard M, AlHamad B and Fink JB. Performance Comparisons of Jet and Mesh Nebulizers Using Different Interfaces in Simulated Spontaneously Breathing Adults and Children. Journal of aerosol medicine and pulmonary drug delivery. 2014.
12.Dugernier J, Reychler G, Depoortere V, Roeseler J, Michotte JB, Laterre PF, Jamar F and Hesse M. Tomoscintigraphiccomparison of lung deposition with a vibrating-mesh and a jet nebulizer. ERS conference poster 2015.
13.Hickin S, Mac Loughlin R, Sweeney L, Tatham A and Gidwani S. Comparison of mesh nebuliser versus jet nebuliser in simulated adults with chronic obstructive pulmonary disease. Poster at the College of Emergency Medicine Clinical Excellence Conference. 2014
14.Page References 1 DMG Halpin , GJ Criner, A Papi, D Singh , A Anzueto , FJ Martinez, AA Agusti , CF Vogelmeier on behalf of the GOLD Science Committee.
15.Global Initiative for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease: The 2020 GOLD Science Committee Report on COVID-19 & COPD https://www.atsjournals.org/doi/pdf/10.1164/rccm.202009-3533SO 2 Ari A.
16.Practical strategies for a safe and effective delivery of aerosolized medications to patients with COVID-19. Respir. Med. 2020; 167. doi:10.1016/j.rmed.2020.105987.
17.Miller A, Epstein D. Safe bronchodilator treatment in mechanically ventilated COVID-19 patients: A single center experience. J. Crit. Care. 2020; 58: 56–57.
18.Respiratory care committee of Chinese Thoracic Society. [Expert consensus on preventing nosocomial transmission during respiratory care for critically ill patients infected by 2019 novel coronavirus pneumonia]. Zhonghua Jie He He Hu Xi Za Zhi 2020; 17: E020.
19.Aerogen Internal data on file, Aug 2020.
20.Fink JB, Ehrmann S, Li J, Dailey P, McKiernan P, Darquenne C et al. Reducing Aerosol-Related Risk of Transmission in the Era of COVID-19: An Interim Guidance Endorsed by the International Society of Aerosols in Medicine. J Aerosol Med Pulm Drug Deliv 2020; : jamp.2020.1615.
21.O’Toole C, McGrath JA, Joyce M, Bennett G, Byrne MA, MacLoughlin R. Fugitive Aerosol Therapy Emissions during Mechanical Ventilation: In Vitro Assessment of the Effect of Tidal Volume and Use of Protective Filters. Aerosol Air Qual Res 2020; 20. doi:10.4209/aaqr.2020.04.0176.
22.Dugernier J, Reychler G, Wittebole X, Roeseler J, Depoortere V, Sottiaux T et al. Aerosol delivery with two ventilation modes during mechanical ventilation: a randomized study. Ann Intensive Care 2016; 6: 73.
23.MacIntyre NR, Silver RM, Miller CW, Schuler F, Coleman RE. Aerosol delivery in intubated, mechanically ventilated patients. Crit Care Med 1985; 13: 81–84.
24.Cinesi Gómez C, Peñuelas Rodríguez Ó, Luján Torné M, Egea Santaolalla C, Masa Jiménez JF, García Fernández J et al. Clinical consensus recommendations regarding non-invasive respiratory support in the adult patient with acute respiratory failure secondary to SARS-CoV-2 infection. Med Intensiva (English Ed 2020; 44: 429–438.
25.Swarnakar R, Gupta N, Halder I, Khilnani G. ICS guidance for nebulization during the COVID-19 pandemic. Lung India 2020; 0: 0.
26.https://www.aarc.org/wp-content/uploads/2020/03/guidance-document-SARS-COVID19.pdf (accessed 13 Nov2020).